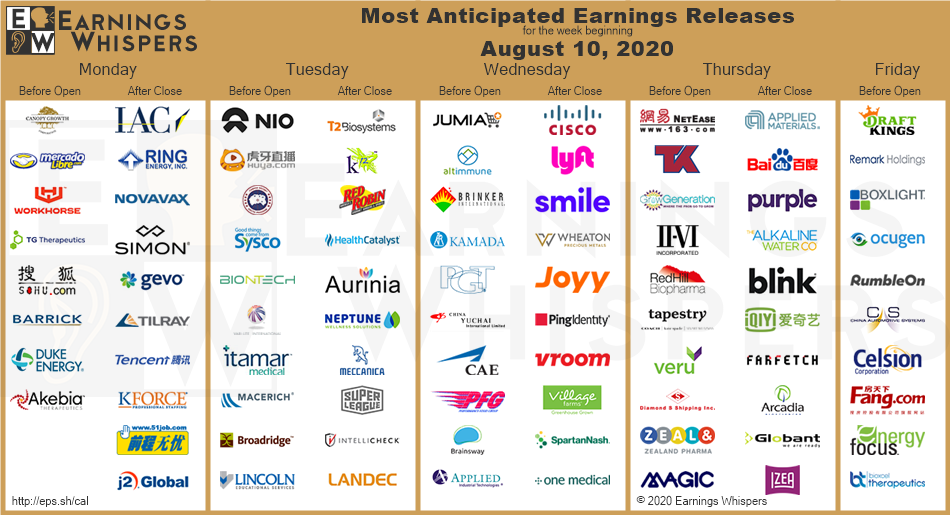
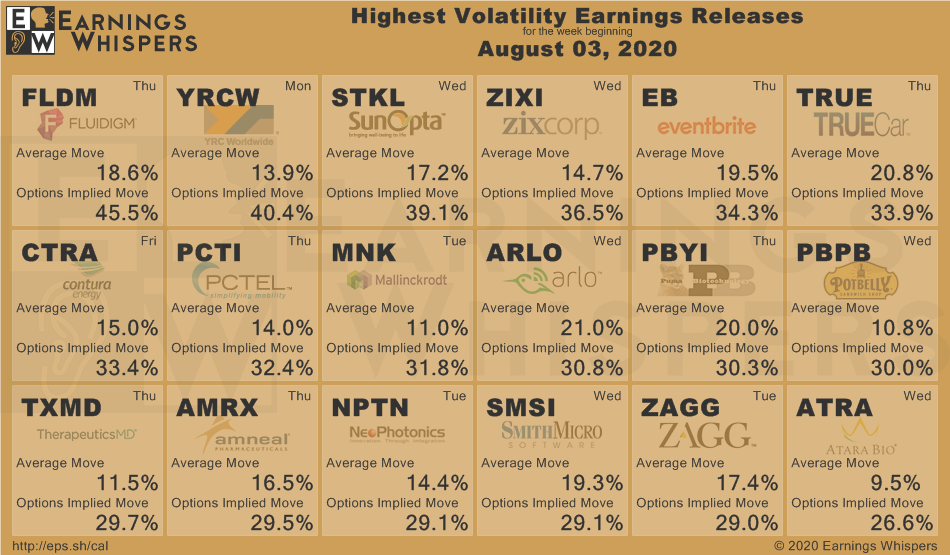
Week of Aug 22, 2020 Weekly Recap & The Week Ahead
Monday, August 24th, 2020‘They know that overstaying the festivities — that is, continuing to speculate in companies that have gigantic valuations relative to the cash they are likely to generate in the future — will eventually bring on pumpkins and mice. But they nevertheless hate to miss a single minute of what is one helluva party. Therefore, the giddy participants all plan to leave just seconds before midnight. There’s a problem, though: They are dancing in a room in which the clocks have no hands.’ — Warren Buffett
1. U.S. Further Tightens Huawei Restrictions — the Commerce Department said that it was restricting Chinese tech-giant Huawei’s access to foreign chips made using U.S. technology on the grounds that it could be used to spy on Americans. The new rule requires non-U.S. firms to get a special license to sell any chips, including off-the-shelf chips, made using U.S. technology to Huawei. A previous rule, imposed by the Trump administration on Huawei in May, was aimed largely at limiting the ability of Huawei’s HiSilicon unit from importing chips made by Taiwan Semiconductor Manufacturing using American chip-making technology. Huawei has already said it would stop making its top-end cellphone chip in mid-September because it can’t get the components. The new rule, which takes effect on Sept. 15, will further curtail Huawei’s ability to source parts for its products.
2. Democrats Nominate Joe Biden in Virtual Roll Call — Democrats formally made Joe Biden their presidential nominee, a role he has sought for more than three decades, capping a come-from-behind victory in the primaries he secured with a broad coalition of his party’s voters. The program included remarks from former Presidents Bill Clinton and Jimmy Carter, and former Secretary of State John Kerry, who was also the Democratic presidential nominee in 2004. “Our party is united in offering you a very different choice: a go-to-work president,” Clinton said.
3. Regeneron and Roche Team Up on Antibody Treatment — Regeneron Pharmaceuticals is partnering with rival drug maker Roche Holding to develop and manufacture Regeneron’s experimental Covid-19 antibody treatment. The treatment, which has not yet been approved by the Food and Drug Administration, is based on a cocktail of two antibodies that neutralize the virus in a manner similar to the way HIV and hepatitis C drug cocktails work. The cocktail is being tested as a treatment for people already infected with the virus and as a prophylactic for those likely to be exposed to the virus, such as front-line health care workers. It is currently in final Phase II/III human trials. In related news, the FDA has delayed an emergency use authorization for a Covid-19 treatment that uses blood plasma of recovered Covid-19 patients after top health officials concluded that clinical trials failed to show it worked.
4. FDA Authorizes Convalescent Plasma for Covid-19 Use — the FDA called an emergency-use authorization, permits use of the treatment on hospitalized Covid-19 patients. For Covid-19 patients and the doctors who treat them, the designation opens up the possibility for faster and easier access to a promising treatment. But the FDA said more clinical studies are necessary for definitive proof of the therapy’s effectiveness. Doctors have been looking for validated coronavirus treatments. Until now, only one other drug, remdesivir from Gilead Sciences Inc., has the FDA’s emergency-use OK. Hospitalized patients who received the plasma within three days of diagnosis, are under the age of 80 and not on mechanical ventilation, benefited the most, with a 35% improvement in survival 30 days after receiving the transfusion compared with patients who got plasma with low antibody levels, according to Dr. Marks.
The week ahead — Economic data from Econoday.com:
href=”http://s317550275.onlinehome.us/Blog/admin/2020/08/24/week-of-aug-22-2020-weekly-recap-the-week-ahead/earnings_weekaug282020-2/” rel=”attachment wp-att-11766″>